Metrology Basics
Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
- product type
- eLearning
- Training type
- eLearning
- Level
- 1
Overview about guideline 21 CFR Part 11
Code of Federal Regulation of Food and Drug Administration (FDA)
- description of several approaches to reduce measurements errors
- correct interpretation of single section in 21 CFR Part 11.
- specific description of requirements of 21 CFR Part 11 when applied to a process or application
Duration: approx. 1 hour
Product note
ATTENTION! Contents of this eLearning only available in English language.
Recently Viewed
Training Guideline FDA 21 CFR Part 11, eLearning
600033-0201-904
1.173,00 DKKexcl. VAT
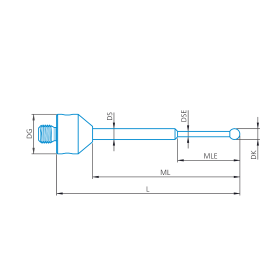
M5, Styli stepped, ruby sphere, tungsten carbide shaft
626115-0106-032
1.344,00 DKKexcl. VAT
Expected soon
Adapter plate VAST XXT TL4, with extension and cone receiver
626103-8434-076
2.817,00 DKKexcl. VAT
Made to Order
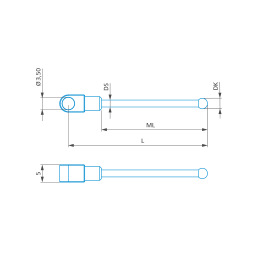
1/4-star stylus M3 XXT, DK3 L40
626103-0302-040
816,00 DKKexcl. VAT
Available